|
Radioactive isotopes (radionuclides to be more correct) are thermodynamically able to change into another nuclide, this decay has to be an exothermic event. Plutonium-239 has a half-life of 24,000 years. Strontium-90 and cesium-137 have half-lives of about 30 years (half the radioactivity will decay in 30 years). Some isotopes decay in hours or even minutes, but others decay very slowly. Radioactive isotopes eventually decay, or disintegrate, to harmless materials. Transuranic wastes, sometimes called TRU, account for most of the radioactive hazard remaining in high-level waste after 1,000 years. These heavier-than-uranium, or "transuranic," elements do not produce nearly the amount of heat or penetrating radiation that fission products do, but they take much longer to decay. These atoms form heavier elements such as plutonium. Second, some uranium atoms capture neutrons produced during fission. These isotopes, called "fission products," account for most of the heat and penetrating radiation in high-level waste. The fission creates radioactive isotopes of lighter elements such as cesium-137 and strontium-90. First, uranium atoms split, creating energy that is used to produce electricity. Then after the fission process, what is left behind includes isotopes of strontium, caesium, and the aforementioned plutonium, which are the main contributors to the hazardous radioactivity of the waste.ĭuring the fission process, two things happen to the uranium in the fuel. It must be enriched in the more radioactive but more fissionable (and thus better energy source) uranium-235, leaving behind the more stable uranium-238, or uranium-238 is converted to more useful fuels such as plutonium-239. First off, uranium as it occurs in nature is not really nuclear fuel. 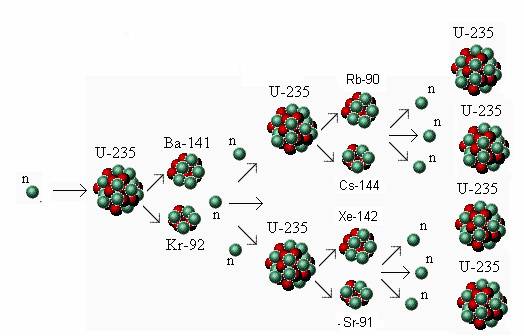
The radioactivity associated with nuclear waste does not come from naturally occurring uranium, but from products associated with processing and using uranium. 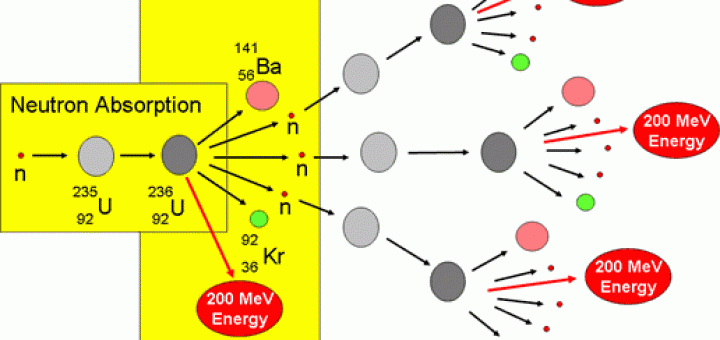
TLDR: How come nuclear power plants release so much radiation when uranium is so relatively stable? That would be an equivalent to $1.4$ tons of pure uranium with $0$ theoretical power loss all directly channeled through retroreflectors and a focusing array all with 100% efficiency, channeling all the alpha decay into a single point onto the detector. Representing with the expression $m(0.5)^$ an hour at Fukushima reactor 1, an approximate of $240$ per day. Its widespread use in wars had many concerned about long-term health effects.Natural uranium has a half life of 4.5 billion years. This has been employed as containers to transport radioactive material, industrial radiography equipment, as well as military uses such as armor plating and armor-piercing projectiles. The leftover from the enrichment process is the creation of Depleted uranium, which has less Uranium-235. This is known as critical mass and enriched uranium usually has between 3 and 5 percent. But you need to have enough of it in your fuel rod. This latter isotope is very good at creating a nuclear chain reaction which makes it possible to have a steady and sustained reaction. Over 99.2 percent of uranium mined on Earth is Uranium-238, with Uranium-235 making almost all the rest. In either scenario, uranium as it is commonly found is not ideal in many reactors. The steam pushes turbines attached to a generator, creating electricity - and, crucially, doesn't produce greenhouse gas emissions. In nuclear power plants, the radioactive emission of fuel rods made of uranium heats up a coolant which in turn is used used to heat up the water in another container and turn it into steam. This ability to store such energy and release it in a well-understood way allowed the use of this element in nuclear power plants.Įqually, the sudden and explosive release of energy from uranium was employed in the atomic bombs. One kilogram of Uranium-235 if led through complete fission is equivalent to the chemical energy that can be extracted by burning 1.5 million kilograms of coal. 
It is the work of Otto Hahn and Fritz Strassmann, that show that Uranium can break into lighter element and Lise Meitner and her nephew Otto Robert Frisch explain and named the process of nuclear fission.Īnd this is what makes Uranium a game-changer, for better or worse. Four decades later, in 1934 a team of Italian physicists led by Enrico Fermi bombarded uranium with neutrons discovering that it emits electrons and positrons. Uranium’s use is no longer in coloring glasses and glazes but in its radioactive properties, discovered by Henri Becquerel in 1896. That accomplishment was achieved by Chemist Eugène-Melchior Péligot. It took until 1841 for the first pure sample of Uranium to be isolated. He named the new element after the planet Uranus, discovered just eight years before by Willaim Herschel. When heated with charcoal it reduce to a black powder, which Kalproth erroneously believed to be pure uranium but it was most likely an oxide.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
